Copper is a reddish-orange metal that is plentiful in nature. Copper ore can be found in many different ore minerals, among them chalcopyrite mineral, chalcocite mineral, covellite mineral, bornite mineral, azurite mineral, dioptase mineral, tennantite mineral, chrysocolla mineral, and digenite mineral. About half of the copper ore used currently comes from chalcopyrite ore. Read More…
Our company may have started small, but we quickly grew to handle the largest aluminum orders. Our metals are made from the highest quality materials, and we pledge to offer superior service to all our customers. We don’t want customers who use us one time, we want to establish lifelong relationships with customers that last generations. Contact us for more info!
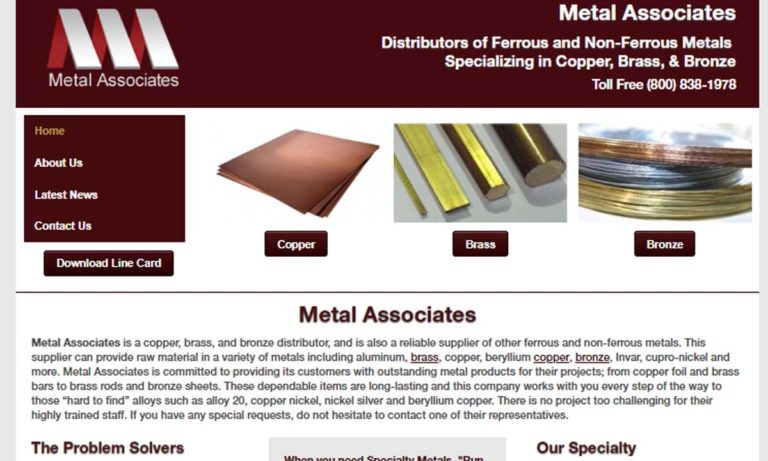
With over 25 years of metallurgy experience we guarantee we can produce exactly what you are looking for. We offer fast delivery and a multitude of beryllium copper sizes. Our highly knowledge staff takes great pride in creating these outstanding copper alloys. We have a reputation of manufacturing the best copper fabrications. Our company has risen in the standard in copper processing. Contact...
At Source 21, Inc., we take pride in offering top-notch copper products to meet a wide range of industrial and commercial needs. When it comes to copper, we've established ourselves as a reliable provider known for quality, accessibility, and customer satisfaction. Our extensive array of copper products is designed to cater to your diverse requirements while ensuring uncompromised excellence.
Schorr Metals is a complete metal service center and your best source for copper and brass extrusions, sheets and more with customized fabrication services, cutting, welding and rolling services. Combining our inventory of copper, brass, steel and other metals with our exceptional customer service and capabilities, the answer is clear - Schorr Metals is your choice for quality, service and value.

We are committed to our customers. Our engineers manufacture copper products that exceed regulatory standards. Our products are the best quality and adhere to a multitude of rigorous physical testing to ensure the reliability of our copper. You can rest assured that you are receiving the best copper. From tubes, rounds, hollows, squares, hexagons and more, we have it all! Please visit our website ...

More Copper Suppliers
Copper, designated by the chemical symbol Cu, is an element situated in Group 11 of the periodic table and bears the atomic number 29. Renowned for its distinctive properties, pure copper is characterized by its softness, malleability, ductility, and nonmagnetic nature. Moreover, it excels as a conductor of both electricity and heat.
One of copper’s remarkable qualities is its ability to form alloys, significantly enhancing its versatility. Notable examples include bronze (composed of copper and tin, CuSn) and brass (composed of copper and zinc, Cu3Zn2). Through alloying, copper can adopt the properties of other metals, broadening its range of applications and making it indispensable in various industrial and technological fields.
Applications of Copper
Copper’s unique properties and ease of availability have made it a preferred choice in numerous industrial and commercial fields, such as electronics, marine manufacturing, wiring, construction, automotive, architecture, power generation, lighting, telecommunications, and food processing.
Copper and its alloys are primarily used for thermal and electrical conductivity, fluid transfer, antimicrobial applications, power transmission, and architectural elements. Furthermore, copper is crucial in vehicle components, transportation systems, refrigeration, and air conditioning.
History of Copper
Early copper working processes included coldworking, annealing, smelting, and lost-wax casting. Humans first discovered copper and later its alloy, bronze, around 5500 BC in the Balkans. Approximately 2000 years later, people began alloying copper themselves. Brass, a copper and zinc alloy, was discovered much later during the Greek period and favored by the Romans.
Ancient societies across present-day Iran, Turkey, Israel, Egypt, India, Greece, and Rome utilized copper and its alloys for various items like coins, weapons, jewelry, and architecture. The Egyptians were particularly prolific, crafting tools, household items, and even makeup from copper. Despite the discovery of iron and subsequent eras, copper remained significant, with major mining operations like the Falun Mine in Sweden starting around 1000 AD and greatly contributing to European copper supply through the 1600s.
The 1700s saw copper used in European shipbuilding, and by the 1800s, advancements like powder metallurgy and electroplating emerged. In 1949, the Finnish company Outokumpu revolutionized copper smelting with the energy-efficient flash smelting process. Recognizing copper’s essential role in human health, the EPA set limits on copper levels in drinking water in 1991. Today, copper’s uses continue to expand, especially in telecommunications and electronics, due to its superconductivity, affordability, and non-sparking nature.
Copper Products
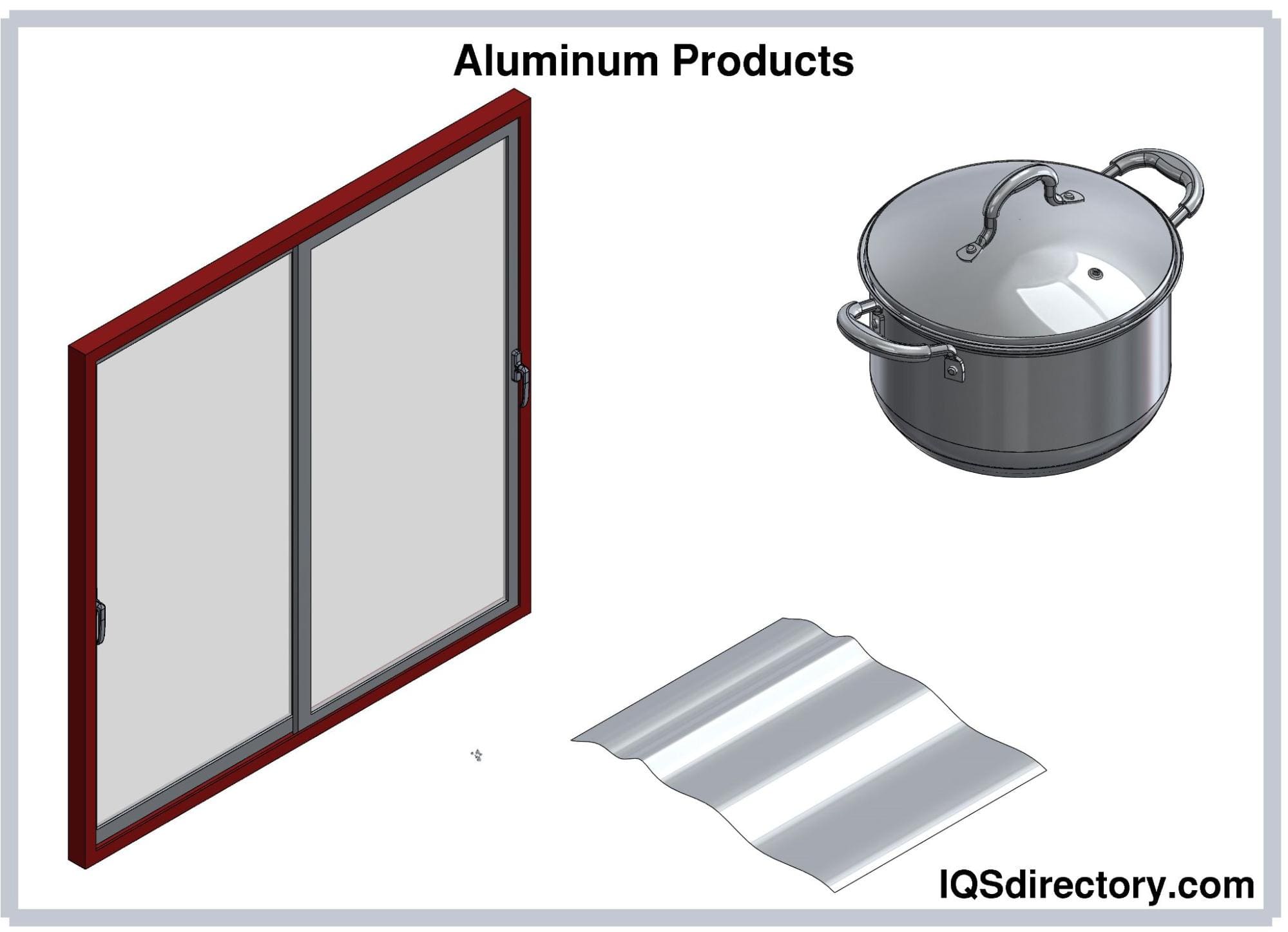
Manufacturers utilize basic copper and copper alloy shape products, such as copper strip and sheet copper, copper foil, copper braid, copper pipes, copper tubes, brass plates, and copper coil, across various industries. They use copper strip and sheet as flat, flexible rolls for roofing and copper foil and braid in electrical and industrial applications. Copper tubes, valued for their corrosion resistance and high heat transfer capabilities, play a crucial role in HVAC and plumbing systems. Brass plates, available as thin, solid square or rectangular pieces, and copper coils, known for their excellent electrical and thermal conductivity, extend the utility of these materials. Fabricated from these shapes, products include microchips, piping systems, copper cookware, propellers, hardware, tools, lighting fixtures, door handles, fasteners, small springs, roofing materials, coins, electrical connectors, wires, adaptors, and valves, demonstrating their versatility and essential role in modern manufacturing and engineering.
Benefits of Copper
Copper, brass, bronze, and other copper alloys provide numerous advantages for manufacturers and users. Firstly, copper and its alloys are non-sparking, meaning they do not produce sparks or fires when subjected to friction. This makes them ideal for use in hazardous environments where combustible vapors, gases, and flammable dust are present, such as oil rigs. Additionally, in culinary applications, copper offers superior heat control compared to stainless steel.
Beyond their practical benefits, copper and copper alloys are also prized for their aesthetic appeal. Their clean, attractive color and finish make them popular in architectural design, both indoors and outdoors. Various surface treatments can enhance their appearance, giving copper an antique or aged look.
Another significant advantage of copper is it’s recyclable. It is one of Earth’s few natural resources that has not been excessively mined. The amount of copper recycled each year nearly matches the amount mined, ensuring its continued abundance. This sustainability, combined with its durability and low maintenance requirements, makes copper a highly favorable material.
Choosing the Right Manufacturer
If you are in need of copper products, it is essential to collaborate with a supplier proficient in both high-quality fabrication and copper extraction. Avoid suppliers who offer impure copper products. To assist you, we have listed several trusted copper alloy manufacturers. Review their profiles to find the right services for your needs.










 Alloy Suppliers
Alloy Suppliers Aluminum
Aluminum Aluminum Extrusions
Aluminum Extrusions Copper-Brass-Bronze
Copper-Brass-Bronze Magnets
Magnets Nickel
Nickel Stainless Steel
Stainless Steel Stainless Steel Tubing
Stainless Steel Tubing Steel Service Centers
Steel Service Centers Titanium
Titanium Tungsten
Tungsten Wire Rope
Wire Rope Castings & Forgings
Castings & Forgings Bulk Material Handling
Bulk Material Handling Electrical & Electronic Components
Electrical & Electronic Components Flow Instrumentation
Flow Instrumentation Hardware
Hardware Material Handling Equipment
Material Handling Equipment Metal Cutting Services
Metal Cutting Services Metal Forming Services
Metal Forming Services Metal Suppliers
Metal Suppliers Motion Control Products
Motion Control Products Plant & Facility Equipment
Plant & Facility Equipment Plant & Facility Supplies
Plant & Facility Supplies Plastic Molding Processes
Plastic Molding Processes Pumps & Valves
Pumps & Valves Recycling Equipment
Recycling Equipment Rubber Products & Services
Rubber Products & Services